by Tyler Irving
Chemistry subject editor
In my last post, I discussed how chemistry is the key to nanotechnology, a hot new field that’s promising to improve everything from smart phones to health care. I also mentioned that nano-sized particles are smaller than the wavelengths of light, which makes it maddeningly difficult to “see” what’s going on down there. Why, if someone could solve that problem, they could practically win the Nobel Prize! A couple of weeks ago, that’s exactly what happened.
To get a Canadian perspective on this, I called up Scott Irvine. A native of Alberta, Irvine is presently a Defence Scientist at Defence Research and Development Canada, but he was a post-doctoral fellow at the Max Planck Institute for Biophysical Chemistry in Göttingen, Germany from 2006 to 2008. His boss was Stefan Hell, one of the three scientists who shared this year’s Nobel Prize in Chemistry. I asked Irvine for some more details on the discovery and what it was like to work with a Nobel Prize winner. Here’s an edited version of our conversation.
___

Scott Irvine, an Alberta native who worked in the lab of Stefan Hell, one of the three scientists who shared this year’s Nobel Prize in Chemistry (Photo credit: Max Planck Institute for Biophysical Chemistry).
What was the problem you were trying to solve?
If you’re using a light microscope, there’s something called a diffraction limit. Basically, because light is a wave, you can only resolve objects down to a certain size, and that size is related to the length of the waves. For visible light, the waves are about 400-500 nanometres apart, so the diffraction limit is half that, about 200 nanometres. So if light can only be focused to a spot size of about 200 nm, trying to see objects or detail separated by amounts less than that is impossible; the image will be a blurred version of the underlying detail.
But I’ve seen microscope images of things smaller than 200 nanometres. Does that mean these images weren’t taken with visible light?
You can use x-rays, which have shorter wavelengths, or electrons, which also have wave-like properties. The problem with that is that both of these can have very high energy, which can be rough on whatever you’re trying to image. If you’re looking at a metal or rock crystal, you might be OK, but if you want to look at a cell, a protein, or some biological molecule that’s very delicate, those methods might destroy it.
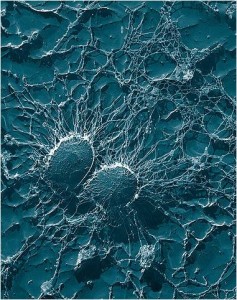
This transmission electron microscope (TEM) image of two Staphylococcus aureus cells shows features smaller than 200 nanometres, but the cells had to be coated in a thin layer of metal first. STED microscopy is a gentler method for imaging tiny features; the technology earned the 2014 Nobel Prize in Chemistry. (Image credit: Eric Erbe, Christopher Pooley, US Department of Agriculture, via Wikimedia Commons)
How do you get past the diffraction limit?
It’s a multi-step process. First, you use a fluorophore, a kind of dye molecule that absorbs light at one wavelength and re-emits it at another wavelength. These dyes are commonly used in biology, and people use a beam of laser light – an excitation beam – to excite them and make them glow.
What Dr. Hell did in the mid 1990s was surround this excitation beam with another beam. The second beam – called the stimulated emission beam – is shaped kind of like a doughnut, with the excitation beam going through the hole. The stimulated emission beam carries away all the fluorescence except for the tiny pinprick at the centre. It basically restricts the fluorescence to a very narrow point in space that you can sweep back and forth over the surface. By scanning across your sample and combining all light from each point in a computer, you can get a much sharper image.
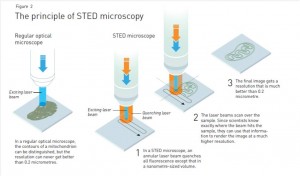
Stimulated emission depletion (STED) microscopy uses one beam of light (orange) to restrict the size of another beam of light to a tiny pinprick (blue). By scanning this pinprick across a sample, scientists can build up a picture of the tiniest objects, like the components within individual cells. Image from “The Nobel Prize in Chemistry 2014 – Popular Information”, via Nobelprize.org
So this is a way of looking at tiny things that’s gentler than using x-ray or electron microscopes?
Exactly. So now you can peer deeper into fine biological structures. For example, you could look at the individual components that make up a cell, or proteins moving around inside it. You could even look at these things while the cell is still alive.
How did you end up working on this technology?
I studied physics at the University of Lethbridge, then did a PhD in electrical engineering at the University of Alberta. I learned a lot about nanotechnology and optics, but I wanted to learn more about how they could be applied to biology problems. I remember doing an internet search for nano, photonics and biology, and Stefan Hell’s lab was the first hit. When I got a post-doctoral fellowship from NSERC, I gave him a call.
Was it hard to have to move to Germany?
At first it was very hard; I remember going to the grocery store, and having to play charades to ask for a bag to carry my groceries in. But I eventually figured it out. The lab was amazing; in my previous lab I was with a handful of people, but now there were 40 or 50 people there. Everyone was great and very intelligent. My wife and I had twins while we were over there, and it was great to have a support network of all these people from all over the world, many with small children of their own.
What kind of research did you do for Hell?
We were trying to improve the technique, to see just how small we could focus the light. I ended up making images of tiny defects in diamond crystals: they act just like fluorophores, but they can stand up to higher-energy lasers. We had a paper in Nature Photonics where we got the resolution below 6 nanometres. That was a world record at the time.
Were you surprised when Hell won the Nobel Prize?
When I was there, it seemed like every other week he was getting another award. He’s a brilliant guy and a very hard worker, so I wasn’t surprised at the idea that he would win it someday. It’s not just one or two papers that win the Nobel Prize; it’s about dedicating your life to a certain area of science and making your impact there, breakthrough after breakthrough. What is surprising to me is that he got it so soon. I thought it would happen, but maybe in 10 years or so.
Lasers and light are all physics stuff, and the problem you’re trying to solve – imaging cells and cell components – is all biology. So how is it that this physical solution to a biological problem wins the Nobel Prize in Chemistry?
Chemistry was an integral component to the STED process. But more importantly, I don’t think any field should take priority over others. We’re all here just trying to figure stuff out. I think that people should just work on whatever they think is interesting, and if that carries them into the neighbouring fields of science, then why not? I’ve heard it said that the really great breakthroughs are made at the interface between sciences. Where one leaves off and another begins, that’s where there’s lots of really cool stuff going on.




