Ainslie Butler, Health, Medicine & Veterinary Sciences editor
A recent report in Science described a new cancer screening blood test, CancerSEEK, that promises to dramatically improve rates of cancer diagnosis. This screening tool, developed at the Johns Hopkins University School of Medicine, is called a ‘liquid biopsy’ and it tests for eight common types of cancer by looking for DNA mutations and unusual proteins in blood.
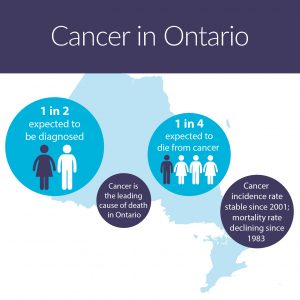
One in two people in Ontario are expected to be diagnosed with cancer in their lifetime. Source: Cancer Care Ontario. Ontario Cancer Statistics 2018. Toronto: Cancer Care Ontario; 2018 – CC
What could this mean for Canadians? One in two Canadians are expected to be diagnosed with cancer, and one in four will die from it, according to the recently released Ontario Cancer Statistics and Canadian Cancer Statistics reports. Slight increases in the rate of newly diagnosed cancer cases have been seen across Canada as a whole, especially among women, while in Ontario, the rate has remained steady for nearly 15 years. Although the rates of cancer diagnosis and survival still vary by region, age, and sex, cancer mortality has been declining overall since the ‘80s. One reason for that overall decline is the steady improvement of diagnostic testing.
As you might expect, diagnostic testing plays a major role in determining the rates of cancer diagnosis across Canada. Advances in medical research have led to better, faster, more effective, and (usually) cheaper tests that can detect specific cancers earlier than ever. Most people are familiar with cancer screening tests such as colonoscopies and mammograms; colorectal cancer and breast cancer are among the most commonly diagnosed cancers, but they also top the list of cancers with the highest mortality rates.
The bottom line is, the more types of cancer we can test for, the more likely we are to catch them, particularly in their early stages. This is where CancerSEEK comes in. The preliminary results for CancerSEEK match or exceed the successful detection rates of similar cancer screening tools. Even better, this blood test is a simple, non-invasive method for detecting several types of cancer, such as ovarian, liver, stomach, pancreatic and esophageal cancers, for which we do not currently have routine screening practices.
Blood testing. Source: Pixabay.com, CC0
If approved, one of the greatest advantages of CancerSEEK over other screening practices would be its relative simplicity. A 2014 survey of Canadian households reported that nearly half of eligible people were not up-to-date for colorectal cancer screening. Visiting specialists and undergoing invasive procedures can be huge barriers to people completing recommended cancer screening. Some procedures, such as colonoscopies, can require fasting, purging and/or sedation, which can be both daunting and disruptive.
For people living in rural communities, there may not be any local specialists, and even for those living in urban environments, getting to their offices and clinics can be challenging. Further, screening capacity varies across Canada, and some Canadians may face long wait times to see specialists even when they are considered at-risk.
Researchers tested CancerSEEK on more than 1,000 patients diagnosed with cancer and found that it successfully diagnosed about 70% of cancer cases (sensitivity). The effectiveness of this procedure at detecting cancers varied among different types and stages of cancer. For example, it successfully detected 100% of early stage liver cancers, but only 20% of early stage esophageal cancers.
The CancerSEEK test performed best at identifying later stage cancers, which makes sense since they typically involve more DNA damage and release higher levels of the abnormal proteins that this test looks for. But, catching cancers in their early stages provides huge opportunities for prevention and treatment, and improves patients’ chances of survival.
Among 800 healthy controls, CancerSEEK incorrectly identified cancer in less than 1% of tests (specificity). This is on par with the specificity of other cancer screening tests such as the Pap smear; however, some researchers fear that CancerSEEK might result in a higher rate of false positive test results in the general population. False positive cancer screening results can lead to unnecessary referrals for additional testing or treatment, and can lead to stress and financial strain and can have impacts on mental health.
CancerSEEK is still in the trial stages, and a trial following 10,000 people over five years is currently underway. There are strict and lengthy processes, both in Canada and internationally, for regulatory approval of new medical procedures. They ensure that new medical technologies are safe and effective. Regulators must determine whether the benefits of the new processes outweigh the risks , and compare them to existing tools to evaluate their relative effectiveness.
If and when CancerSEEK or other similar ‘liquid biopsy’ tools become available in Canada, getting blood drawn at your healthcare provider’s office and sent off to the lab will be an easy and hassle-free way to catch cancer early and increase your chances of survival.
~30~
Feature image: Could a new blood test change the Canadian cancer landscape? Source: qimono, CC0